
You’ll have to calculate moles and multiply it by 6.02*10 23 to find atoms. How many molecules in a mole?Ī mole contains 6.02214076×10 23 numbers of molecules. A mole depends on both mass and molecular mass. 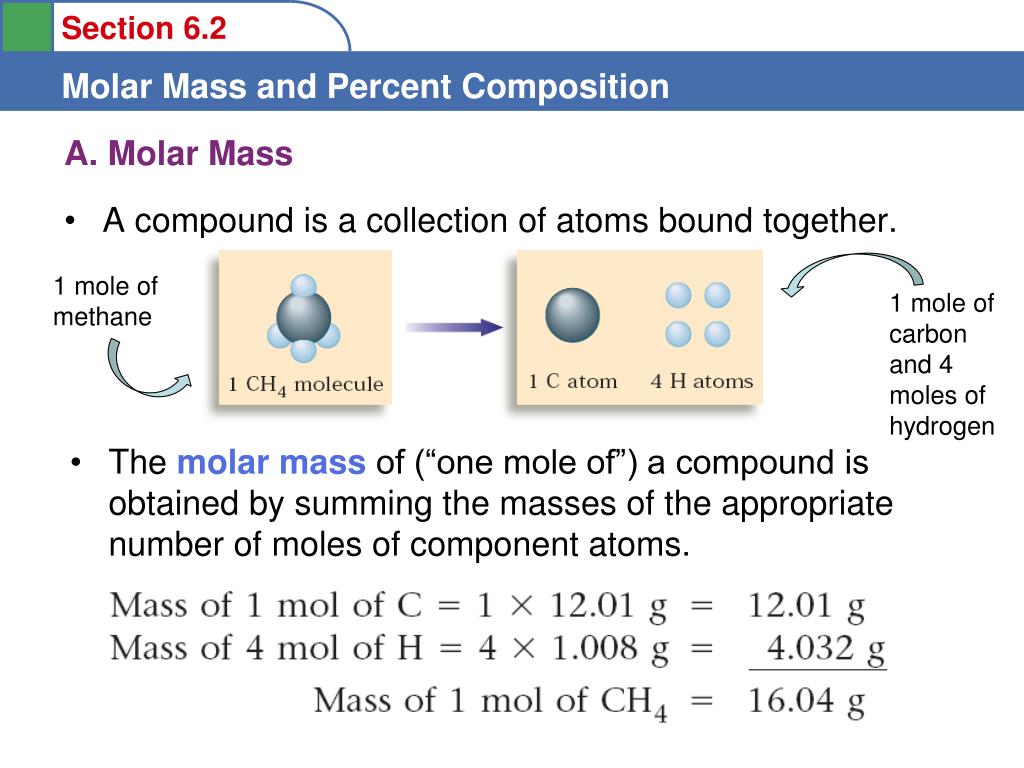
Each element or substance has different mass and molecular mass. One mole can have varying quantity in grams depending on the element. To find the molar mass of gaseous compounds, use our molar mass of gas calculator. Use moles to grams converter to verify the number of moles in the above example. Step 2: Apply formula as shown below or place the values in moles to atoms calculator. ∴ in one molecule of oxygen there are 2 atom. Step 1: Write down and identify the values. How many moles are in 16 grams of Oxygen gas? On the other hand, if you are interested in mole calculation without using atoms to grams calculator, follow the examples below. One mole of NaCl has 02 × 10 23Na and 6.02 × 10 23 Cl ions.Ĭonverting grams to moles is super easy if you use mass to moles calculator above.One mole of oxygen is equal to has 02 × 10 23 molecules.One mole of helium gas is equal to 02 × 10 23atoms.This number is also referred to as Avogadro’s number. Wondering how much is a mole? A mole of an element or molecule contains exactly 6.02214076×10 23 particles. It is used to measure substances in small quantities. Mole is an SI unit for the amount of a substance. In this space, we will cover mole definition, moles formula, how to convert grams to moles without using grams to moles calculator, and some examples to convert mass to moles, etc. Chem., 1981, 256, 12-14.Are you struggling with questions like how to convert moles to grams, how to find moles, or how to find moles from grams? Well, don’t worry! We have got it sorted out for you.īy using this moles to grams calculator, you can convert moles to mass and grams, grams to mass and moles, and last but not least mass to moles and grams. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN), and Nomenclature Commission of IUB (NC-IUB).Newsletter 1981, Arch. However, NC-IUB and JCBN believe that the dalton is valuable they know that it is widely used in biochemistry and support its continued use.ġ. The Comité international des poids et mesures did not accept the request. l for length, A for absorbance, v for rate of reaction. Symbols for quantities, unlike those for units, are italicized when derived from the Latin alphabet (some quantities are symbolized by Greek letters), e.g. Symbols for units based on proper names do begin with capital letters, e.g. dalton, not Dalton, as with volt, joule, etc. Names of units do not begin with capital letters. Since molecular mass is a mass, no special symbol for the quantity other than m is needed. It is 1.6605655 x 10 -27 kg within about 6 parts per million.ģ. Its relation to the kilogram therefore depends on experimental determination and cannot be stated exactly. The dalton is one twelfth of the mass of the nuclide 12C. The numerical value of the molecular mass in daltons is equal to the numerical value of the molar mass ( M m or M) in g mol -1 and to the relative molecular mass ( M r).Ģ. a 144-Da fragment, a 15-kilodalton molecule, a 2.6-MDa ribosome.ġ. The name 'dalton' and symbol 'Da' would allow adjectival use, e.g. Its symbol (u) is strange in that this one unit should be symbolized with a 'u' for unit, and some of its multiples and submultiples, e.g. The word 'dalton' is already used in the biochemical literature. In asking other IUPAC bodies their views on this proposal, the Interdivisional Committee for Nomenclature and Symbols states that it finds merit in the proposal and adds the following arguments and notes.ġ. 
NC-IUB and JCBN are therefore asking IUB and IUPAC to apply to the Comité international des poids et mesures (International Committee of Weights and Measures) to "approve the name 'dalton' (symbol: Da) as an alternative to the name 'atomic' mass unit' (symbol: u)". The third form of expression may sometimes be useful, or would be if the name of the unit and its symbol were more convenient. Although M r is the most used, M is sometimes needed, as in the Svedberg equation M = R. These are all correct ways of saying the same thing by present IUPAC recommendations: Manual of Symbols and Terminology for Physicochemical Quantities and Units, 1979, Pergamon, Oxford also in Pure Appl. The molar mass (symbol: M) of glucose is 180 g mol -1 the relative molecular mass (symbol: M r) of glucose is 180 the molecular mass of glucose is 180 atomic mass units (symbol: u). Masses of moles Masses of molesFrom the JCBN/NC-IUB Newsletter 1981
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
